Vocal communication is ubiquitous in the mammalian kingdom and astounding in its richness and diversity. While vocal behaviors in different animals may be highly specialized, fundamental systems of information processing in the brain are preserved across species. I believe in the potential of a comparative, cross-species approach for illuminating how fundamental properties of vocal production and processing have evolved - in humans, as well as in our near and distant cousins.
My research focuses on vocal plasticity in echolocating bats (Carollia perspicillata) and the neural mechanisms that jointly support the processing of auditory inputs and executive control of vocalization.
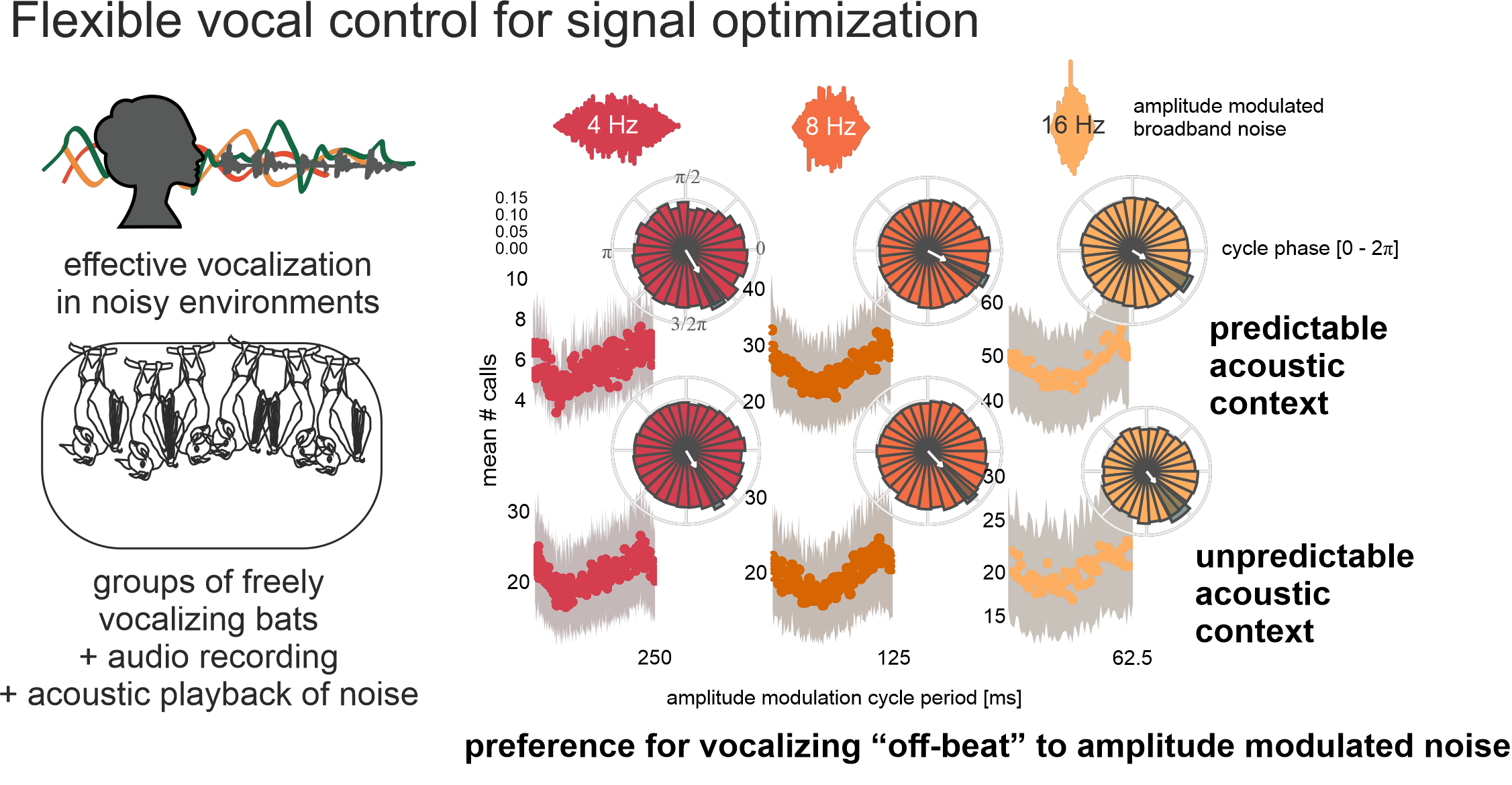
Study 1: Talking in a noisy world
The natural world is full of noise, which can interfere with intra-specific acoustic communication. Many bats, including our model species Carollia, rely heavily on the returning echos of their biosonar pulses for navigation, while also engaging in rich vocal interactions. How do bats deal with acoustic jamming from their auditory environment?
In a pair of behavioral experiments, we showed that bats actively avoid acoustic interference from background noise by seeking out temporal “windows of of opportunity” in ambient noise. By vocalizing more often in periods of low-amplitude noise, they are able to maintain the signal integrity of their echolocation pulses or social calls.
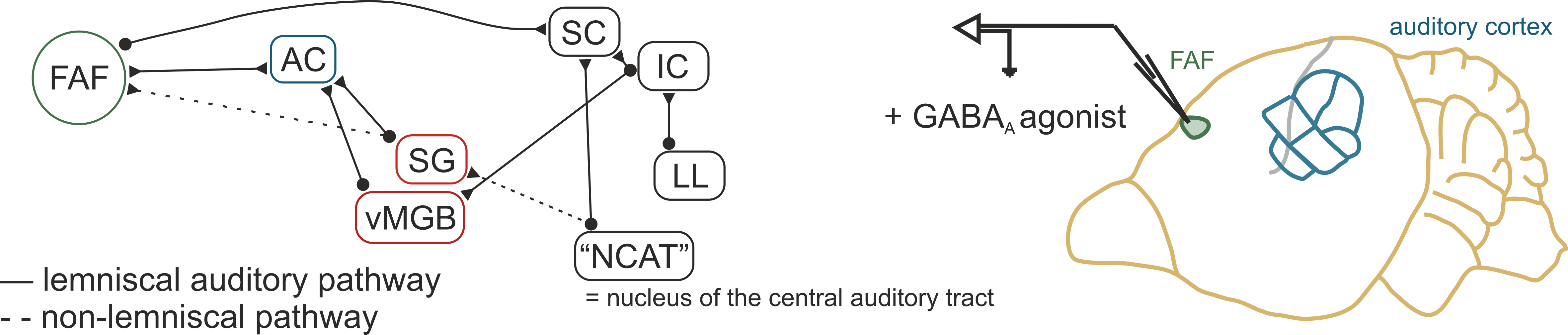
Study 2: Investigating a frontal locus of auditory processing and vocal control
The frontal cortex of Carollia bats features a small region, the fronal auditory field (FAF), believed to be involved in integrating auditory information and the motor execution of vocalizations.
To probe the role of this region in both auditory processing and vocal production, we record extracellular activity in vivo from awake animals during vocalization and during playback of different acoustic stimuli after administering the GABA agonist muscimol, which reversibly inhibits FAF.
Study 3: Turn taking in a vocal [learning?] animal
Carollia is a species of the family Phyllostomidae, which features several vocally talented bat species. Carollia pups are not born with a fixed and unchanging vocal repertoire. Rather, their vocal abilities develop in the course of infancy.
In this study, we examine whether Carollia pups and mothers engage in in systematic turn-taking, where communication calls are emitted in turns and listeners wait until the current speaker has stopped calling to begin their turn. Turn-taking is believed to underlie complex social interactions and to be an evolutionary precursor of human language, but has not yet been observed in this species.